It’s time for our next 14-day moving average determinations for SARS-CoV-2 for the United States and my thoughts on vaccines, SARS-CoV-2 therapeutic agents and mutant viruses. We use the WORLDOMETERS aggregators data set to make any projections since it includes data from the Department of Veterans Affairs, the U.S. Military, federal prisons and the Navajo Nation.
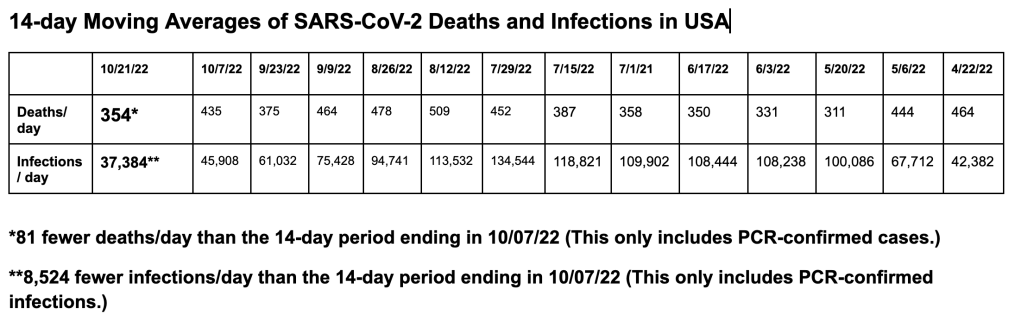
SARS-CoV-2 infections per day in the United States have decreased for the fourth time in 12 weeks; however, there is still widespread underreporting by states, a failure to capture positive home tests, and a decreased PCR screening program in most states. Deaths per day in the United States have decreased by 81 deaths per day; however, many states are not reporting deaths in a timely manner. The number of infections per day has decreased by 8,524. The CDC estimates that BA.5 accounted for 62.2% (a 17% drop from 10/7/22), BQ.1 accounted for 9.4%, BQ.1.1 accounted for 7.2%, BA.4.6 accounted for 11.3%, BF.7 accounted for 6.7%, BA.2.75 accounted for 1.6%, BA.2.27.2 accounted for 1.3%, and BA.4 accounted for 0.4%, in the week ending October 22.
The total percentage of BQ variant infections in the region that includes New York and New Jersey is 28.4%.
The total percentage of BQ variant infections in the region that includes Pennsylvania, Delaware, Maryland, West Virginia, and Virginia is 17.8%.
The total percentage of BQ variant infections in the region that includes California, Nevada, Arizona, and Hawaii is 13.6%.
Data on the rapid spread of a dangerous variant category, the BQ variants, was withheld by the CDC in their weekly reports until last week. The data on BQ.1 and BQ.1.1, the last being a variant with five significant spike protein mutations leading to escape from immunity from prior infections or vaccination. In addition, our monoclonal antibody therapies do not work for these isolates. Infections and hospitalizations in New York are rapidly increasing, secondary to BQ variants. We can expect this pattern to continue in many states, since New York has been a harbinger of things to come throughout the pandemic.
These emerging BQ variants are descendants of BA.5. It’s troubling that, despite the availability of a BA.5 bivalent SARS-CoV-2 vaccine, few people are getting vaccinated. As of October 19, the CDC reports that 19.4 million people have received the bivalent vaccine. That’s only 8.5% of people who received the primary series and 5% of the overall population.
There has been no new UK Health Security Agency Technical Briefing since October 7. The October 7 Technical Briefing says, “From UK data, BQ.X, BA.2.75.2 and BF.7 are the most concerning variants in terms of both growth and neutralisation data at present; there is also supportive animal model data for BA.2.75.”
| Variant | Sublineage of | Spike Mutations | Global Sequences | UK Sequences |
| BF.7 | BA.5.2.1 | R346T | 9,809 (1,752 from Belgium) | 663 |
| BQ.1.1 | BA.5 | N460KK444TR346T | 326(20 countries) | 60 |
| BJ.1 | BA.5 | 13 non- synonymous spike mutations, 7 in RBD and including4 predicted immune escape locations | 123 (10 countries, most cases in India) | 1 |
| BS.1 | BA.2.3.2 | R346TL452RN460KG476S | 25 (15 from Japan) | 0 |
In Monterey County, as of 10/22/22, 2.1% of 0-4 year-olds and 40.1% of 5-11 year-olds have received the first two doses of vaccine, while 73.3% of 12-17 year-olds have received two doses. Only 54.1% of Monterey County residents have received a third dose of the vaccine. The Monterey County Health Department does not publish data on how many residents have received the new BA.5 bivalent booster vaccine. On June 17, The FDA authorized both the Pfizer and Moderna vaccines for use in children ages 6 months to four years. We believe children under 5 should be vaccinated as soon as possible. All Monterey County residents should get up to date on COVID-19 vaccinations, including the bivalent BA.5 booster, as soon as possible.
On 10/21/22, the United States had 19,652 documented new infections. There were also 190 deaths. Thirty-four states did not report their infections, and 36 states didn’t report their deaths. In the United States the number of hospitalized patients has decreased slightly (-1% compared to the previous 14 days) in many areas and was 26,810 on October 22. On 10/21/22 there were 2,707 patients who are seriously or critically ill; that number was 2,753 two weeks ago. The number of critically ill patients has decreased only by 47 in the last 14 days, while at least 4,951 new deaths occurred. The number of critically ill patients has decreased for the fifth time in twenty-nine 14-day periods. Patients are still dying each day (average 354/day). Omicron BA.4, BA.4.6, BA.5, and BF.7 variants are still causing infections. A new variant BQ.1.1, descended from Omicron BA.5, is causing increasing numbers of infections and hospitalizations in New York. In Singapore a different Omicron BA.2 variant, XBB, is causing rapidly increasing infections. Past infections with a BA.1, BA.2 or BA.5 variants will not prevent infections with any of the newer variants.
As of 10/21/22, we have had 1,092,606 deaths and 99,055,537 SARS-CoV-2 infections in the United States. We have had 532,369 new infections in the last 14 days. We are adding an average of 266,185 new infections every seven days. For the pandemic in the United States we are averaging one death for every 90.65 infections or over 11,030 deaths for each one million infections. As of 10/21/22, thirty-eight states have had greater than 500,000 total infections, and 38 states have had greater than 5,000 total deaths. Forty-six states have had greater than 2,000 deaths, and 43 states have greater than 2,000 deaths per million population. Seven states have over 4,000 deaths per million population: Mississippi (4,350), Arizona (4,330), Alabama (4,186), West Virginia (4,178), New Mexico (4,111), Tennessee (4,103) and Arkansas (4,114). . Eighteen states (Alabama, Virginia, Missouri, North Carolina, Indiana, Tennessee, Massachusetts, Ohio, Michigan, Georgia, Illinois, New Jersey, Pennsylvania, Florida, Texas, New York, Arizona and California) have had greater than 20,000 deaths. Seven states have had greater than 40,000 deaths: Florida (82,065 deaths), Texas (91,584 deaths), New York (72,694 deaths), Pennsylvania (47,582 deaths), Georgia (40,552 deaths), Ohio (40,111 deaths) and California (96,721 deaths, 20th most deaths in the world).
On 11/20/20, there were 260,331 (cumulative) deaths in the US from SARS-CoV-2. Since 11/20/20 (23 months), there were 825,605 new deaths from SARS-CoV-2. For nineteen of those months, vaccines have been available to all adults. During these eighteen months, 520,509 people have died of SARS-CoV-2 infections. Most of the hospitalizations and deaths could have been prevented by vaccination, proper masking, and social distancing.
As of 10/21/22, California was ranked 33rd in the USA in infection percentage at 28.65%. In California, 24.68% of people were infected in the last 19 months. As of 10/21/22, 24 states have had greater than 30% of their population infected. Fifty states have greater than 20% of their population infected.
Worldwide, average deaths per day are 1,569 for the last 14 days, which is a 97 deaths-per-day increase over the previous 14 days. The United States accounts for 22.56% (354 per day) of all deaths per day in the world over the last two weeks. Worldwide infections per day were 310,532. The United States accounts for 12.04% of those infections (or 37,384 infections per day).
FDA-Approved Oral Drug Treatments for SARS-CoV-2
Pfizer has developed PAXLOVID™, an oral reversible inhibitor of C3-like protease of SARS-CoV-2. The drug inhibits this key enzyme that is crucial for virus production. The compound, also called Compound 6 (PF-07321332), is part of the drug combination PAXLOVID™ (PF-07321332; ritonavir), which just successfully completed a Phase 2-3 trial in humans in multiple countries. The preliminary results were announced on 11/5/21 by Pfizer. The results show that 89% of the hospitalizations and deaths were prevented in the drug treatment arm. The drug was administered twice a day for five days. No deaths occurred in the treatment group, and ten deaths occurred in the placebo group. The study was stopped by an independent data safety monitoring board, and the FDA concurred with this decision. Pfizer applied for an Emergency Use Authorization for this drug on 11/15/21. This drug was approved on 12/23/21. We have only been able to obtain PAXLOVID™ for two patients who we successfully treated with this drug obtained from CVS in Salinas (East Alisal Street; phone number 831-424-0026). They were expecting another shipment on 1/28/22. In my opinion, this agent, if more widely available, could markedly alter the course of every coronavirus infection throughout the world.
Merck has developed the oral drug Molnupiravir, which induces RNA mutagenesis by viral RNA-dependent RNA polymerase of SARS-CoV-2 and other viruses. According to Kabinger et al, “Viral RNA-dependent RNA polymerase uses the active form of Molnupiravir, β-D-N4-hydroxycytidine triphosphate, as a substrate instead of cytidine triphosphate or uridine triphosphate. When the RNA-dependent RNA polymerase uses the resulting RNA as a template, β-D-N4-hydroxycytidine triphosphate directs incorporation of either guanine or adenine, leading to mutated (viral) RNA products. Analysis of RNA-dependent RNA polymerase–RNA complexes that contain mutagenesis products has demonstrated that β-D-N4-hydroxycytidine (the active form of Molnupiravir) can form stable base pairs with either guanine or adenine in RNA-dependent RNA polymerase explaining how the polymerase escapes proofreading and synthesizes mutated RNA” (quotation modified for clarity). The results of the phase 3 trial of Molnupiravir were published in the NEJM article “Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients” by Angélica Jayk Bernal, M.D. et al. (December 16, 2021 DOI: 10.1056/NEJMoa2116044). In this phase 3 study in the Molnupiravir group, 28 patients were hospitalized and one death occurred. In the placebo group, 53 patients were hospitalized and 9 died. Overall, 47% of hospitalizations and deaths were prevented by Molnupiravir. If you do a post hoc analysis and just look at deaths, Molnupiravir would prevent 89% of deaths. An Emergency Use Authorization by the FDA for Molnupiravir was approved on 12/24/21.The dose of Molnupiravir approved is four 200 mg capsules orally twice a day for five days. Diarrhea is reportedly a side effect in two percent of patients. I treated my first patient with Molnupiravir on 1/28/22. Currently more Molnupiravir is available weekly in the United States than PAXLOVID™ (see chart below; data from PHE.gov). Locally Molnupiravir is still available at CVS in Monterey (Fremont Blvd.; phone number: 831-375-5135) and CVS in Salinas (East Alisal Street; phone number 831-424-0026).
28, p740–746 (2021)with four 200 mg capsules orally twice a day for five dayfour 200 mg capsules orally twice a day for five daysIntravenous Drug Treatment for non-hospitalized SARS-CoV-2 Infected Patient
FDA-Approved Intravenous Monoclonal Antibody Treatment for Non-Hospitalized SARS-CoV-2 Patients
Bebtelovimab is a monoclonal antibody treatment for mild-to-moderate COVID-19 in adults and pediatric patients (12 years of age and older weighing at least 40 kg) with positive results of direct SARS-CoV-2 viral testing, and who are at high risk for progression to severe COVID-19, including hospitalization or death, and for whom alternative COVID-19 treatment options approved or authorized by FDA are not accessible or clinically appropriate. The authorized dose of bebtelovimab is 175 mg, given as an intravenous injection over at least 30 seconds. The U.S. Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) for this drug on 2/11/22. Bebtelovimab is a human antibody that demonstrates neutralization against the Omicron variants and is available in every state and many hospitals and some clinics. If you are planning on using a monoclonal antibody to treat a SARS-CoV-2 infection, currently only bebtelovimab has activity against all Omicron variants, including BA.4.6. Researchers at Columbia University recently reported that “The loss of activity of tixagevimab and cilgavimab [components of Evusheld] against BA.4.6 leaves us with bebtelovimab as the only therapeutic mAb that has retained potent activity against all circulating forms of SARS-CoV-2.” For this reason, we no longer recommend Evusheld for immunocompromised patients with Omicron infections.
An examination of the three variants that Wang et al identified as capable of immune escape in patients who receive the two monoclonal antibodies that are contained in Evusheld reveals that all three variants have a mutation in the spike protein at position 346. The changes substitute an uncharged amino acid— threonine (T), serine (S), or isoleucine (I)—for a positively-charged amino acid, arginine. This just goes to show that a single point mutation in the spike protein can render a monoclonal antibody treatment ineffective. Policy makers should keep in mind that the only way to prevent new drug-resistant variants like BA.4.6 from emerging is to prevent transmission of SARS-CoV-2 in the first place, using non-pharmaceutical interventions. The lack of use of N95 masks, with impending winter and influenza outbreaks, on top of COVID-19, is not wise public health and infectious disease policy. If we don’t make effective use of the non-pharmaceutical interventions available to us (masks, ventilation, social distancing), then the pharmaceutical interventions we have will all eventually be useless.
Two virologists collaborated on Twitter to create the figure below, which Professor Johnson titled “Convergent Evolution on Steroids.” It shows the key mutations present in many of the currently-circulating Omicron subvariants and demonstrates that mutation at site 346 is becoming more and more common. This means that even if prevalence of BA.4.6 wanes, we are still likely to have drug resistance issues with other newer variants.
Watching World Data
Over the next few months, we’ll be paying close attention to correlations between the SARS-CoV-2 data, the number of isolates identified in various countries and states, and the non-pharmaceutical interventions (like mask mandates and lockdowns) put in place by state and national governments. Data on infections, deaths, and percent of population infected was compiled from Worldometers. Data for this table for SARS-CoV-2 Isolates Currently Known in Location was compiled from GISAID and the CDC. It’s worth noting that GISAID provided more data than the CDC.
| Location | Total Infections as of 10/21/22 | New Infections on 10/21/22 | Total Deaths | New Deaths on 10/21/22 | % of Pop.Infected | SARS-CoV-2 Isolates Currently Known in Location | National/ State Mask Mandate | Currently in Lockdown |
| World | 632,432,678(4,347,443 new infections in 14 days). | 349,351 | 6,581,530(21,965 new deaths in last 14 days) | 1,189 | 8.11% | B2 lineageAlpha/B.1.1.7 (UK)Eta/B.1.525 (Nigeria/UK)Iota/B.1.526 (USA-NYC)Beta/B.1.351 (SA)Epsilon/B.1.427 + B.1.429 (USA)*Gamma/P.1 (Brazil)Zeta/P.2 (Brazil)A lineage isolateV01.V2 (Tanzania)APTK India VOC 32421Delta/B.1.617.2 (India)BV-1 (Texas, USA)Kappa/B.1.617.1 (India)Lambda/C.37 (Peru)Theta/P.3 (Philippines) Mu/B.1.621 (Colombia)C.1.2 (South Africa 2% of isolates in July 2021)R1 (Japan)Omicron/B.1.1.529 + BA.1 + BA.2 + BA.3 (South Africa November 2021)B.1.640.1 (Congo/France)B.1.640.2 (Cameroon/France)Four new recombinants 12/31 to 3/22)BA.2.12.1 (USA)BA.4 (South Africa)BA.5 (South Africa)BA.2.75 (India 7/22)BA.4.6 (USA 7/22)BF.7BJ.1XBBBQ.1BQ.1.1BS.1 | No | No |
| USA | 99,055,537(ranked #1) 532,369 new infections in the last 14 days. | 19,652(ranked #7) 34 states failed to report infections on 10/21/22. | 1,092,606(ranked #1) 4,951 new deaths reported in the last 14 days. | 190 36 states failed to report deaths on 10/21/22. | 29.58% | B2 lineageAlpha/B.1.1.7 (UK)Eta/B.1.525 (Nigeria/UK)Iota/B.1.526 (USA-NYC)Beta/B.1.351 (SA)Epsilon/B.1.427 + B.1.429 (USA)*Gamma/P.1 (Brazil)Zeta/P.2 (Brazil)Delta/B.1.617.2 (India)BV-1 (Texas, USA)Theta/P.3 (Philippines) Theta/P.3 (Philippines) Kappa/B.1.617.1 (India)Lambda/C.37 (Peru)Mu/B.1.621 (Colombia)R1(Japan) Omicron/B.1.1.529 + BA.1 + BA.2 (South Africa November 2021)B.1.640.1 (Congo/France)Recombinant Delta AY.119.2- Omicron BA.1.1 (Tennessee, USA 12/31/21)\BA.2BA.2.12.1 (United States)BA.4 (South Africa 11/21)BA.5 (South Africa 11/21)BA.2.75 (India 7/22)BA.4.6 (USA 7/22) | No | No |
| Brazil | 34,822,174(ranked #4) 64,917 new infections in the last 14 days. | 3,400 | 687,581(ranked #2; 732 new deaths in 14 days) | 37 | 16.16% | B2 lineageAlpha/B.1.1.7 (UK)Beta/B.1.351 (SA)Gamma/P.1 (Brazil)Zeta/P.2 (Brazil)Lambda/C.37 (Peru)Mu/B.1.621 (Colombia) Omicron/B.1.1.529 + BA.1 (South Africa November 2021)BA.2BA.2.12.1 (United States)BA.4 (South Africa 11/21)BA.5 (South Africa 11/21) | No | No |
| India | 44,640,748(ranked #2); 31,991 new infections in 2 weeks. | 2,112 | 528,957(ranked #3) 199 new deaths in 2 weeks. | 4 | 3.17% | B2 lineageAlpha/B.1.1.7 (UK)Beta/B.1.351 (SA)Gamma/P.1 (Brazil)Epsilon/B.1.427 + B.1.429 (USA)*Eta/B.1.525 (Nigeria/UK)APTK India VOI 32421Delta/B.1.617.2 (India)Kappa/B.1.617.1 (India)Iota/B.1.526 (USA-NYC) Omicron/B.1.1.529 + BA.1 (South Africa November 2021)B.1.640.1 (Congo/France)BA.4 (South Africa 11/21)BA.5 (South Africa 11/21)BA.2.75 (India) | No | No |
| United Kingdom | 23,855,522(ranked #7) 120,249 new infections in 2 weeks. | – | 192,682 (ranked #7) 1,794 new deaths in 2 weeks | – | 34.82% | B2 lineageAlpha/B.1.1.7 (UK)Eta/B.1.525 (Nigeria/UK)Beta/B.1.351 (SA)Epsilon/B.1.427 + B.1.429 (USA)*Gamma/P.1 (Brazil)Delta/B.1.617.2 (India)Theta/P.3 (Philippines) Kappa/B.1.617.1 (India)Lambda/C.37 (Peru)Mu/B.1.621 (Colombia)C.1.2 (South Africa)Omicron/B.1.1.529 + BA.1 (South Africa November 2021)B.1.640.1 (Congo/France)XD (AY.4/BA.1) recombinantXF (Delta/BA.1) recombinantXE (BA.1/BA.2) recombinantBA.2BA.2.12.1 (United States)BA.4 (South Africa 11/21)BA.5 (South Africa 11/21)BA.2.75 (India 7/22) | No | No |
| California, USA | 11,332,345(ranked #14 in the world; 39,798 new infections in the last 14 days). | 998 | 96,721 (ranked #20 in world) 303 new deaths in the last 14 days | 14 | 28.65% | B2 lineageAlpha/B.1.1.7 (UK)Eta/B.1.525 (Nigeria/UK)Beta/B.1.351 (SA)Gamma/P.1 (Brazil)Epsilon/B.1.427 + B.1.429 (USA)*Zeta/P.2 (Brazil)Delta/B.1.617.2 (India)Theta/P.3 (Philippines) Kappa/B.1.617.1 (India)Lambda/C.37 (Peru) Mu/B.1.621 (Colombia) Omicron/B.1.1.529 + BA.1 (South Africa November 2021)BA.2BA.2.12.1 (United States)BA.4 (South Africa 11/21)BA.5 (South Africa 11/21)BA.2.75 (India 7/22) | No | No |
| Mexico | 7,106018(ranked #19) 4,587 new infections in 14 days). | 453 | 330,321(ranked #5)181 new deaths in 14 days) | 15 | 5.40% | No | No | |
| South Africa | 4,025,375(ranked #37; 4,587 new infections in 14 days). | 416 | 102,257 (ranked #18) 63 new deaths in 14 days) | 11 | 6.62% | B2 lineageAlpha/B.1.1.7 (UK)Beta/B.1.351 (SA)Delta/B.1.617.2 (India)Kappa/B.1.617.1 (India) C.1.2 (South Africa, July 2021)Omicron/B.1.1.529 + BA.1 (South Africa November 2021)B.1.640.1 (Congo/France)BA.2BA.4 (South Africa 11/21)BA.5 (South Africa 11/21) | No | No |
| Canada | 4,314,718(ranked #33) 43,827 new infections in 14 days). | 3,067 | 46,025(ranked #25)631 new deaths in the last 14 days | 49 | 11.23% | No | No | |
| Poland | 6,333,591 (ranked #21; 22,629 new infections in 14 days). | 1,170 | 118,015 (ranked #15)272 new deaths in the last 14 days | 24 | 16.78% | B2 lineageAlpha/B.1.1.7 (UK)Eta/B.1.525 (Nigeria/UK)Beta/B.1.351 (SA)Delta/B.1.617.2 (India)Mu/B.1.621 (Colombia)Omicron/B.1.1.529 + BA.1 + (South Africa November 2021),Omicron/B.1.1.529 +BA.3 | No | No |
| Russia | 21,354,915(ranked #10), 190,973 new infections in 14 days). | 9,761 (ranked #8) | 389,359(ranked #4)1,368 new deaths in 14 days | 93 | 14.64% | No | No | |
| Peru | 4,152,019(ranked #32, 3,858 new infections in 14 days). | 314 | 216,877(ranked #6) 177 new deaths in the last 14 days | – | 12.32% | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India)Gamma/P.1 (Brazil)Iota/B.1.526 (USA-NYC)Lambda/C.37 (Peru)Mu/B.1.621 (Colombia)Omicron/B.1.1.529 + BA.1 (South Africa November 2021) | No | No |
| Iran | 7,555,694(ranked #17; 4,672 new infections in last 14 days) | 139 | 144,540(ranked #12) 69 new deaths in the last 14 days | 4 | 8.78% | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India)Beta/B.1.351 (SA)Omicron/B.1.1.529 + BA.1 (South Africa November 2021) | No | No |
| Spain | 13,488,015(ranked #12; 46,047 new infections in 14 days). | 3,630 | 114,858 (ranked #16)390 new deaths in 14 days | 31 | 28.87% | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India)Beta/B.1.351 (SA)Gamma/P.1 (Brazil)Epsilon/B.1.427 + B.1.429 (USA)*Eta/B.1.525 (Nigeria/UK)Iota/B.1.526 (USA-NYC)Kappa/B.1.617.1 (India)Mu/B.1.621 (Colombia)Omicron/B.1.1.529 + BA.1 (South Africa November 2021)B.1.640.1 (Congo/France) | No | No |
| France | 36,524,605 (ranked #3; 758,248 new infections in the last 14 days). | 49,087 (ranked #2) | 156,337 (ranked #10)803 new deaths in 14 days. | 81 | 55.69% a 2.17% increase in 14 days. | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India) Omicron/B.1.1.529 South Africa November 2021)B.1.640.1 (Congo/France)B.1.640.2 (Cameroon/France)GKA (AY.4/BA.1) recombinant | No | No |
| Germany | 35,172,693(ranked #4; 1,224,661 new infections in 14 days.) | 49,087 (ranked #2) | 152,482 (ranked #11)1,947 new deaths in 14 days | 204 | 41.93% 1.71% increase in 14 days | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India) Delta/B.1.617.2 (India) Omicron/B.1.1.529 South Africa November 2021)GKA (AY.4/BA.1) recombinant | No | No |
| South Korea | 25,244,255 (ranked #6 310,499 new infections in 14 days). | 24,709(ranked #8) | 28,952 (ranked #37) 338 new deaths in 14 days | 30 | 49.18% | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India) Delta/B.1.617.2 (India) Omicron/B.1.1.529 South Africa November 2021) | No | No |
| Vietnam | 11,496,354 (ranked #13; 10,933 new infections in 14 days). | 582 | 43,159 (ranked #26) | – | 11.61% | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India) Delta/B.1.617.2 (India) Omicron/B.1.1.529 South Africa November 2021) | No | No |
| Netherlands | 8,494,705 (ranked #16; 33,354 new infections in 14 days). | 952 | 22,683 (ranked #41) | 5 | 49.35% | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India) Delta/B.1.617.2 (India) Omicron/B.1.1.529 South Africa November 2021)GKA (AY.4/BA.1) recombinant | No | No |
| Denmark | 3,131,149(ranked #40) 12,835 new infections in 14 days. | 652 | 7,248 (ranked #79 126 new deaths in the last 14 days) | 13 | 53.66% | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India) Delta/B.1.617.2 (India) Omicron/B.1.1.529 South Africa November 2021)GKA (AY.4/BA.1) recombinant | No | No |
| Taiwan | 7,379,205(ranked #18)569,950 new infections in 14 days | 37,265 (ranked #3) | 12,206 (ranked #59 937 new deaths in the last 14 days) | 78 | 30.89% 2.53% of population has been infected in the last 14 days | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India) Delta/B.1.617.2 (India) Omicron/B.1.1.529 South Africa November 2021) | No | No |
| Japan | 21,926,231(ranked #9)463,493 new infections in the last 14 days | 31,593(ranked #5) | 46,152(ranked #25) 831 new deaths in the last 14 days | 67 | 17.45% | B2 lineageAlpha/B.1.1.7 (UK)Delta/B.1.617.2 (India) Delta/B.1.617.2 (India) Omicron/B.1.1.529 South Africa November 2021)BA.2*BA.5* | No | No |
What Our Team Is Reading This Week
- Effectiveness of Monovalent mRNA Vaccines Against COVID-19–Associated Hospitalization Among Immunocompetent Adults During BA.1/BA.2 and BA.4/BA.5 Predominant Periods of SARS-CoV-2 Omicron Variant in the United States — IVY Network, 18 States, December 26, 2021–August 31, 2022 (MMWR) https://www.cdc.gov/mmwr/volumes/71/wr/mm7142a3.htm
- “Three-dose monovalent mRNA VE estimates against COVID-19–associated hospitalization decreased with time since vaccination. Three-dose VE during the BA.1/BA.2 and BA.4/BA.5 periods was 79% and 60%, respectively, during the initial 120 days after the third dose and decreased to 41% and 29%, respectively, after 120 days from vaccination.”
- Saliva antibody-fingerprint of reactivated latent viruses after mild/asymptomatic COVID-19 is unique in patients with myalgic-encephalomyelitis/chronic fatigue syndrome (Frontiers in Immunology) https://doi.org/10.3389/fimmu.2022.949787
- SARS-CoV-2 Nsp6 causes cardiac defects through MGA/MAX complex-mediated increased glycolysis (Preprint) https://doi.org/10.21203/rs.3.rs-1677754/v1
- WHO’s Therapeutics and COVID-19 Living Guideline on mAbs needs to be reassessed (The Lancet) https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(22)01938-9/fulltext
- SARS-CoV-2 disrupts host epigenetic regulation via histone mimicry (Nature) https://doi.org/10.1038/s41586-022-05282-z
- Real-world effectiveness of molnupiravir and nirmatrelvir plus ritonavir against mortality, hospitalisation, and in-hospital outcomes among community-dwelling, ambulatory patients with confirmed SARS-CoV-2 infection during the omicron wave in Hong Kong: an observational study (The Lancet) https://doi.org/10.1016/S0140-6736(22)01586-0
- An ACE2-dependent Sarbecovirus in Russian bats is resistant to SARS-CoV-2 vaccines (PLOS Pathogens) https://doi.org/10.1371/journal.ppat.1010828
- Plasma cytokine levels reveal deficiencies in IL-8 and gamma interferon in Long-COVID (Preprint) https://doi.org/10.1101/2022.10.03.22280661
- Lingering cardiac involvement in previously well people after mild COVID-19 (Nature Medicine) https://www.nature.com/articles/s41591-022-02002-y
- SARS-CoV-2 variants of concern and variants under investigation in England: technical briefing 46 (UK Health Security Agency) https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1109820/Technical-Briefing-46.pdf
- Rapid initiation of nasal saline irrigation to reduce severity in high-risk COVID+ outpatients (Ear, Nose, and Throat Journal) https://doi.org/10.1177/01455613221123737
- SARS-CoV-2 Omicron boosting induces de novo B cell response in humans (Preprint) https://doi.org/10.1101/2022.09.22.509040
- Imprinted SARS-CoV-2 humoral immunity induces converging Omicron RBD evolution (Preprint) https://www.biorxiv.org/content/10.1101/2022.09.15.507787v2
- Note: This is a frightening paper and if the conclusions are accurate humans in general are in trouble. From other papers the facts that HERV-M and EBV are activated by SARS-CoV-2 infection does not bode well for humans moving forward.
- SARS-CoV-2 Omicron boosting induces de novo B cell response in humans (Preprint) https://www.biorxiv.org/content/10.1101/2022.09.22.509040v1
- Evolution of antibody immunity following Omicron BA.1 breakthrough infection (Preprint) https://www.biorxiv.org/content/10.1101/2022.09.21.508922v1
- SARS-CoV-2 Spike protein promotes vWF secretion and thrombosis via endothelial cytoskeleton-associated protein 4 (CKAP4) (Signal Transduction and Targeted Therapy) https://www.nature.com/articles/s41392-022-01183-9
- Long-term neurologic outcomes of COVID-19 (Nature Medicine) https://www.nature.com/articles/s41591-022-02001-z
- Pulmonary Dysfunction after Pediatric COVID-19 (Radiology) https://pubs.rsna.org/doi/10.1148/radiol.221250
- Severe Respiratory Illnesses Associated with Rhinoviruses and/or Enteroviruses Including EV-D68 – Multistate, 2022 (CDC) https://emergency.cdc.gov/han/2022/han00474.asp
- VACCINE INFORMATION FACT SHEET FOR RECIPIENTS AND CAREGIVERS ABOUT COMIRNATY (COVID-19 VACCINE, mRNA), THE PFIZER-BIONTECH COVID-19 VACCINE, AND THE PFIZER-BIONTECH COVID-19 VACCINE BIVALENT (ORIGINAL AND OMICRON BA.4/BA.5) TO PREVENT CORONAVIRUS DISEASE 2019 (COVID-19) FOR USE IN INDIVIDUALS 12 YEARS OF AGE AND OLDER https://labeling.pfizer.com/ShowLabeling.aspx?id=14472
- Pfizer/BioNTech COVID-19 Omicron-Modified Bivalent Vaccine https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2022-09-01/07-covid-swanson-508.pdf
- Coronavirus (COVID-19) Update: FDA Authorizes Moderna, Pfizer-BioNTech Bivalent COVID-19 Vaccines for Use as a Booster Dose (FDA) https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-moderna-pfizer-biontech-bivalent-covid-19-vaccines-use
- Resistance of SARS-CoV-2 Omicron Subvariant BA.4.6 to Antibody Neutralization (Preprint) https://www.biorxiv.org/content/10.1101/2022.09.05.506628v1.full
- SARS-CoV-2 variants of concern and variants under investigation in England Technical briefing 45, 9 September 2022 (UK Health Security Agency) https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1103869/Technical-Briefing-45-9September2022.pdf
- SARS CoV-2 detected in neonatal stool remote from maternal COVID-19 during pregnancy (Pediatric Research) https://doi.org/10.1038/s41390-022-02266-7
- SARS-CoV-2 variants of concern: spike protein mutational analysis and epitope for broad neutralization (Nature Communications) https://doi.org/10.1038/s41467-022-32262-8
- Is SARS-CoV-2 an oncogenic virus? (Journal of Infection) https://doi.org/10.1016/j.jinf.2022.08.005
- Emergence of immune escape at dominant SARS-CoV-2 killer T cell epitope (Cell) https://doi.org/10.1016/j.cell.2022.07.002
- Neurological and psychiatric risk trajectories after SARS-CoV-2 infection: an analysis of 2-year retrospective cohort studies including 1 284 437 patients (The Lancet Psychiatry) https://doi.org/10.1016/S2215-0366(22)00260-7
- An Antibody from Single Human VH-rearranging Mouse Neutralizes All SARS-CoV-2 Variants Through BA.5 by Inhibiting Membrane Fusion (Science Immunology) https://doi.org/10.1126/sciimmunol.add5446
- Myalgic Encephalomyelitis/Chronic Fatigue Syndrome (ME/CFS) is common in post-acute sequelae of SARS-CoV-2 infection (PASC): Results from a post-COVID-19 multidisciplinary clinic (Preprint) https://doi.org/10.1101/2022.08.03.22278363
- Evaluation of publication bias for 12 clinical trials of molnupiravir to treat SARS-CoV-2 infection in 13,694 patients (Preprint) https://www.researchsquare.com/article/rs-1913200/v1
- Anti-neuronal antibodies against brainstem antigens are associated with COVID-19 (eBioMedicine) https://doi.org/10.1016/j.ebiom.2022.104211
- Risk of SARS-CoV-2 Acquisition in Health Care Workers According to Cumulative Patient Exposure and Preferred Mask Type (JAMA Infectious Diseases) http://jamanetwork.com/article.aspx?doi=10.1001/jamanetworkopen.2022.26816
- Coronavirus spike protein activated natural immune response, damaged heart muscle cells (AHA Basic Cardiovascular Sciences Meeting) https://newsroom.heart.org/news/coronavirus-spike-protein-activated-natural-immune-response-damaged-heart-muscle-cells
- Distinguishing features of Long COVID identified through immune profiling (Preprint) https://doi.org/10.1101/2022.08.09.22278592
- Virological characteristics of the SARS-CoV-2 Omicron BA.2.75 (Preprint) https://doi.org/10.1101/2022.08.07.503115
- Multiple pathways for SARS-CoV-2 resistance to nirmatrelvir (Preprint) https://doi.org/10.1101/2022.08.07.499047
- Transmissible SARS-CoV-2 variants with resistance to clinical protease inhibitors (Preprint) https://doi.org/10.1101/2022.08.07.503099
- Comparative pathogenicity of SARS-CoV-2 Omicron subvariants including BA.1, BA.2, and BA.5 (Preprint) https://doi.org/10.1101/2022.08.05.502758
- Neuropathology and virus in brain of SARS-CoV-2 infected non-human primates (Nature Communications) https://doi.org/10.1038/s41467-022-29440-z
- SARS-CoV-2 Brain Regional Detection, Histopathology, Gene Expression, and Immunomodulatory Changes in Decedents with COVID-19 (Journal of Neuropathology and Experimental Neurology) https://doi.org/10.1093/jnen/nlac056
- Examination of SARS-CoV-2 In-Class Transmission at a Large Urban University With Public Health Mandates Using Epidemiological and Genomic Methodology (JAMA) http://jamanetwork.com/article.aspx?doi=10.1001/jamanetworkopen.2022.25430
- Antiviral treatment is more effective than smallpox vaccination upon lethal monkeypox virus infection (Nature) https://doi.org/10.1038/nature04295
- Impact of Pre-Existing Chronic Viral Infection and Reactivation on the Development of Long COVID (Preprint) https://doi.org/10.1101/2022.06.21.22276660
- Notes from the Field: Increase in Pediatric Intracranial Infections During the COVID-19 Pandemic — Eight Pediatric Hospitals, United States, March 2020–March 2022 (MMWR) http://dx.doi.org/10.15585/mmwr.mm7131a4

You must be logged in to post a comment.